Interventional Care


We notice that you are visiting us from . This site only services US-based visitors. Would you like to visit the site that is appropriate for your location?

As of 30 November 2021, the United States has had 48,525,271 cases of COVID-19 leading to 779,859 deaths. In 2020, COVID-19 was the third leading cause of death in the United States, ranking only behind cardiovascular disease and cancer.1 Likely COVID-19 will also be the third leading cause of death in the United States again in 2021. The University of Washington projects estimates that by 1 March 2022, total deaths in the United States due to COVID-19 will be ~1,024,000. Receipt of a COVID-19 vaccine is the most effective preventive strategy to prevent COVID-19. This blog will review COVID-19 vaccines with a focus on the rationale for booster doses, vaccination of persons who have had COVID-19, and vaccine use in children 5 years and older.
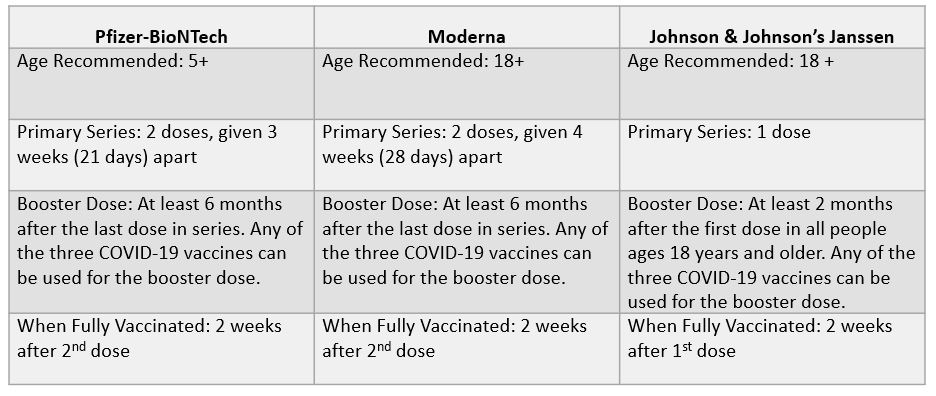
At the present time, there are three COVID-19 vaccines available in the United States either approved or authorized by the U.S. Food and Drug Administration (FDA) and recommended by the U.S. Centers for Disease Control and Prevention (CDC): Pfizer-BioNTeck (available for ages 5+), Moderna (available for ages 18+) and Johnson & Johnson’s Janssen (available for ages 18+).2 Recently, booster doses of Pfizer and Moderna were authorized for all adults by the FDA. The CDC currently recommends that all persons older than 18 years of age receive a booster dose.
Additional details on current CDC COVID-19 vaccine recommendations are summarized in the table below adapted from the CDC.
COVID-19 vaccines have been determined to be both safe and highly effective. Booster doses have been demonstrated to substantially increase the antibody levels in persons who have received a primary vaccine series. Further, two studies from Israel have both demonstrated that within two weeks of receiving a booster, the risk of developing symptomatic COVID-19 and severe COVID-19 is substantially lower. The study by Bar-On and colleagues showed that at least 12 days after the booster dose, the rate of confirmed infection was lower in the booster group than in the non-booster group by a factor of 11.3 (95% confidence interval [CI], 10.4 to 12.3); the rate of severe illness was lower by a factor of 19.5 (95% CI, 12.9 to 29.5).3 The study by Barda and co-workers showed that at least 7 days after a booster dose compared with receiving only two doses at least 5 months ago, was estimated to be 93% for admission to hospital, 92% for severe disease, and 81% (44 vs seven events; 59–97) for COVID-19-related death. Thus, all eligible persons should receive a booster dose.4
Two studies have now demonstrated that persons with natural protection (i.e., persons who have had COVID-19) have a substantially higher risk of reinfection than fully vaccinated persons. Cavanaugh and colleagues using a case-control design reported that Kentucky residents who were not vaccinated had 2.34 times the odds of reinfection compared with those who were fully vaccinated (odds ratio [OR] = 2.34; 95%CI=1.58–3.47).5 Bazio et al. using data collected from 187 hospitals reported that among COVID-19–like illness hospitalizations among adults aged ≥18 years whose previous infection or vaccination occurred 90–179 days earlier, the adjusted odds of laboratory-confirmed COVID-19 among unvaccinated adults with previous SARS-CoV-2 infection were 5.49-fold higher than the odds among fully vaccinated recipients of an mRNA COVID-19 vaccine who had no previous documented infection (95% confidence interval = 2.75–10.99).6 Thus, all persons who have had COVID-19 should receive a COVID-19 vaccine series when no longer infectious.
COVID-19 vaccines are now available for children ages 5 years and older. As with adults, COVID-19 vaccines have been found to be highly effective and safe in children. In recent months, the number and percent of children developing COVID-19 have increased. Although children, in general, have experienced less severe disease than adults, severe hospitalizations and deaths may occur.7 Adolescent hospitalization rates and mortality are substantially higher from COVID-19 compared with influenza.8 As with adults, infected children may develop COVID-19 “long haulers” syndrome with symptoms (e.g., brain fog, fatigue, etc.) lasting months.
Finally, current vaccines, in the era of the Delta variant, remain highly effective in preventing symptomatic COVID-19 and even more effective in preventing hospitalizations and deaths (CDC). Booster doses have been demonstrated to substantially increase the antibody levels in persons who have received a primary vaccine series. The U.S. now ranks 51st in the world in the level of COVID-19 vaccine coverage for its citizens (NY Times). Increasing COVID-19 vaccine uptake is crucial to protecting individuals and reducing the risk of SARS-CoV-2 transmission.
Additional Recommendations for Immunocompromised People:
Stay up-to-date on how COVID-19 vaccinations are trending in the United States here.
References: