Interventional Care


We notice that you are visiting us from . This site only services US-based visitors. Would you like to visit the site that is appropriate for your location?
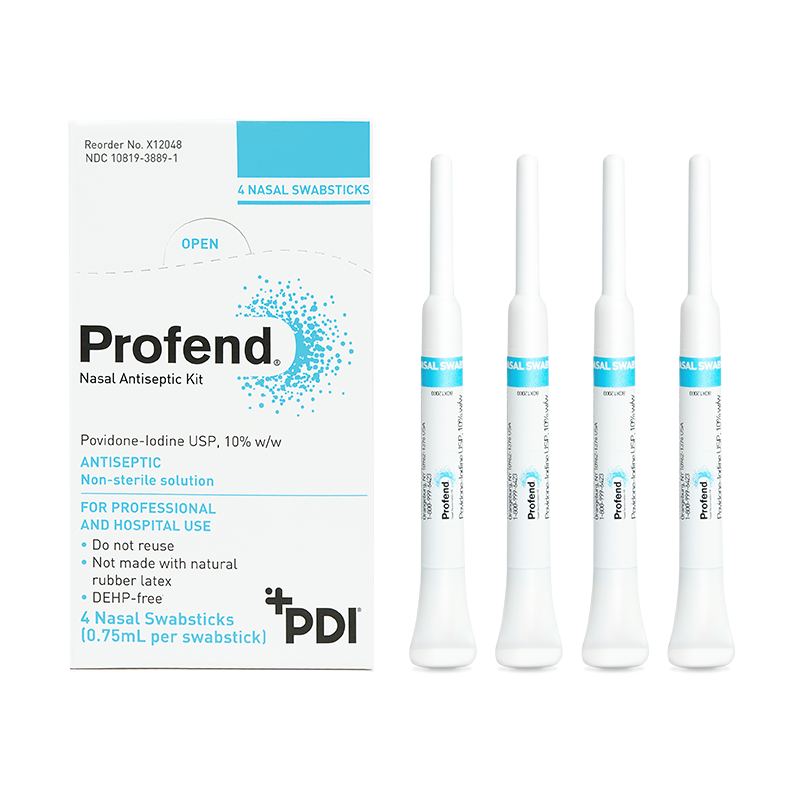

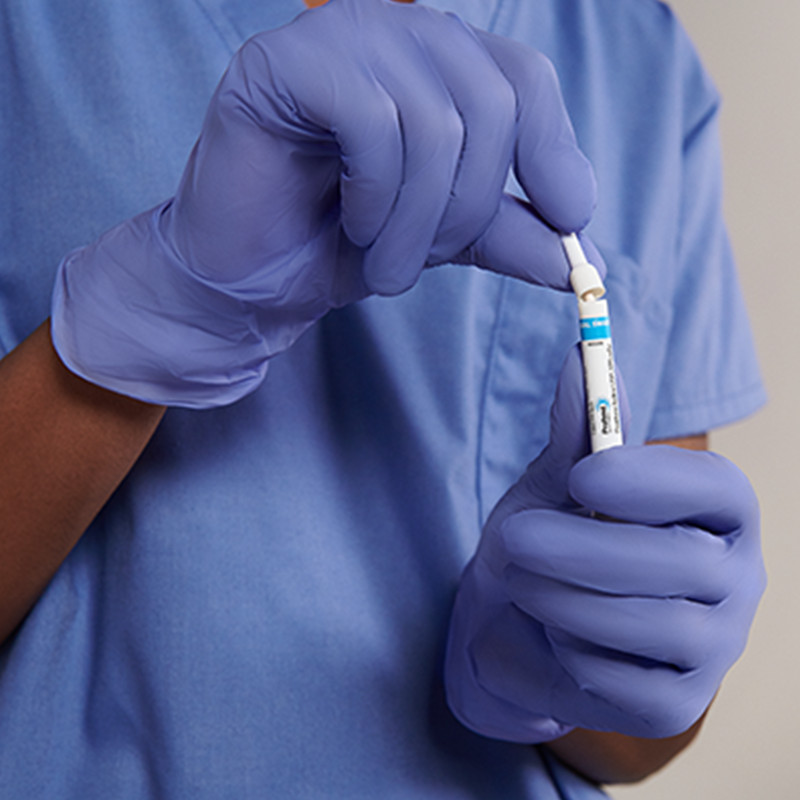
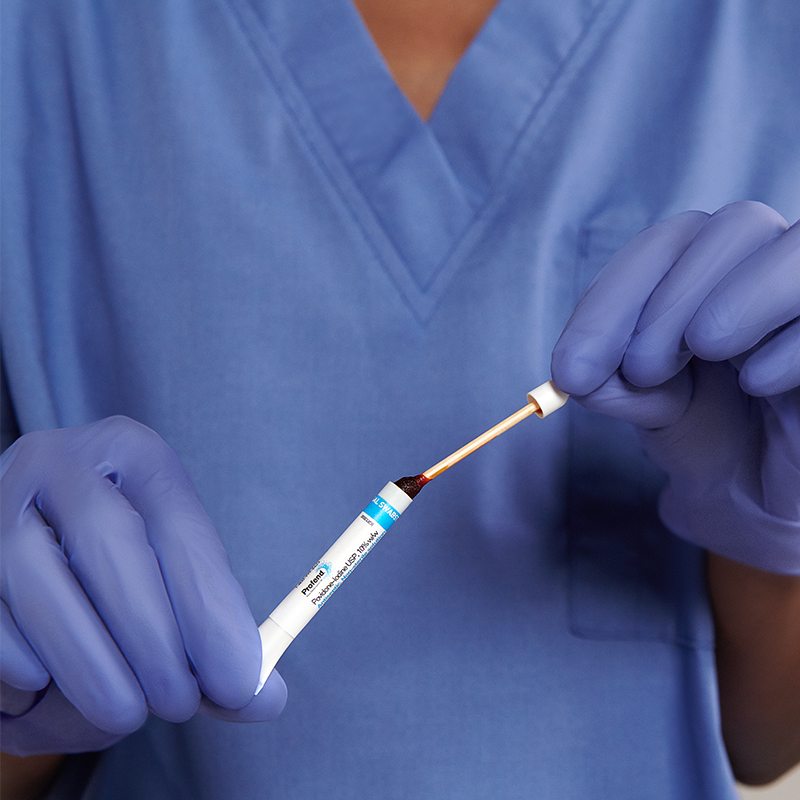
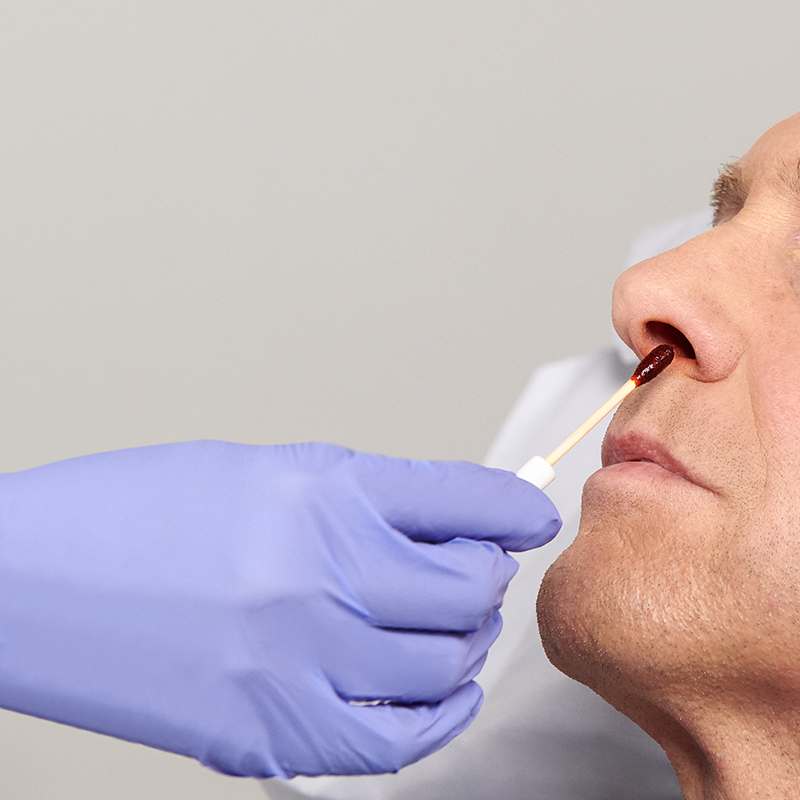

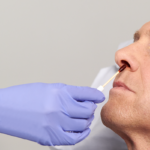

10% Povidone-Iodine (PVP-I) nasal swabsticks used to proactively defend your patients against the risk of infections related to S. aureus.
Want to take prevention further? Through our CATALYST CARE Program, customers gain added support in improving compliance with pre-operative nasal decolonization. Plus, with our money-back guarantee, you can be confident in the process. Learn how to become part of the program today.
SELECT A SIZE
Total treatment time: 60 seconds; 15 seconds per swabstick
Kills 99.7% of S. aureus in 10 minutes.
10% Povidone-Iodine (PVP-I)
Neat, dry-handle for limited mess.
Self-contained, pre-saturated with a unique snap and swab design
Each kit contains 4 pre-saturated swabsticks.
S. aureus-colonized patients in the surgical unit, ICU, and other areas of the hospital
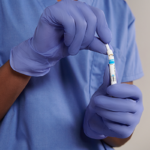
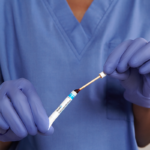
Applied to patient by clinical staff for assured compliance.
In this PDI Call on Clinical episode, Holly Montejano, MS, CIC, CPHQ, VA-BC, sits down with Kimberly Atrubin,…
The recently released, May 2021, AORN guideline for preoperative skin antisepsis describes the importance of nasal and skin…
Learn more about our Profend Nasal Antiseptic Kit, the latest addition to the Interventional Care product portfolio targeting…
Povidone-iodine as an Alternative to Mupirocin for Nasal Decolonization: A New Domain for Antimicrobial Stewardship and Patient Safety…
See how PDI Profend® Nasal Antiseptic Kits made a difference for infection prevention at Hunterdon Medical Center in…
PDI offers a broad range of evidence-based, market-leading Interventional Care, Environment of Care, and Patient Care solutions, all…
This instructions for use sign contains step-by-step directions on how to properly open the applicator tube and for…
How is the Profend® Nasal Antiseptic Kit used?
The Profend Nasal Antiseptic Kit swabsticks are designed to be applied to the circumference of the patient’s nostril and anterior nares. In total, two swabsticks are applied to each nostril for 15 seconds per swabstick. Thus, using 4 swabsticks, the total treatment time is 60 seconds.
What is the Profend® Nasal Antiseptic Kit?
The Profend Nasal Antiseptic Kit is a pack of four swabsticks that are pre-saturated with 10% (w/w) Povidone-Iodine antiseptic solution and are applied to the nostrils/anterior nares of the nose to proactively defend patients against Staphylococcus aureus (S. aureus), other bacteria that can cause Surgical Site Infections (SSIs), and other healthcare-acquired infections (HAIs).
When is the Profend® Nasal Antiseptic Kit used?
The Profend Nasal Antiseptic Kit can be used on patients who have tested positive for S. aureus. As an alternative to a “test and treat” strategy, the ease and speed of application and economical design make Profend suitable for universal decolonization of all patients as well, saving time and money spent on patient testing. Regardless of decolonization strategy, Profend has proven efficacy—in a study of healthy volunteers, the Profend Nasal Antiseptic Kit reduced S. aureus by 99.7% in just 10 minutes.* For pre-operative use, clinicians can choose to apply the product to the patient at least 10 minutes prior to surgery depending on hospital protocol.
*SOURCE: 1 PDI in vivo Study 0113-CTEVO.
Where is the Profend® Nasal Antiseptic Kit used?
The Profend Nasal Antiseptic Kit can be used anywhere in a healthcare facility where there are patients who may be nasally colonized with S. aureus and therefore have an elevated risk of developing an SSI or other HAIs.
Who uses the Profend® Nasal Antiseptic Kit?
Perioperative nurses who care for patients prior to surgery and nursing staff in other areas of the hospital,ie. ICU, are the primary users of the Profend Nasal Antiseptic Kit,. Profend offers unique features valued by clinicians – a preference study showed that over 90% of nurses preferred Profend over other PVP-I nasal antiseptic products.
SOURCE: PDI user acceptance study.
Why is the Profend® Nasal Antiseptic Kit used?
Up to 30% of healthy adults are nasally colonized with S. aureus1. 85% of S. aureus SSIs come from the patient’s own nasal flora.2 Nasal colonization with these bacteria increases the patient risk of developing an SSI by 9 times.3 To help reduce this risk, Povidone-Iodine is used as a broad spectrum antiseptic that has proven efficacy in reducing S. aureus. It offers an added advantage over the widely-used nasal antibiotic, mupirocin, in that there is no known bacterial resistance to PVP-I, thereby supporting antibiotic stewardship within the healthcare environment.
1 VandenBergh MF, Yzerman EP, van Belkum A, Boelens HA, Sijmons M, Verbrugh HA. Follow-up of Staphylococcus aureus nasal carriage after 8 years: redefining the persistent carrier state. J Clin Microbiol. 1999;37:3133–3140.
2 Septimus EJ. Nasal Decolonization: What antimicrobials are more effective prior to surgery? Am J Infect Control 2019;47S:A53-A57. doi: 10.1016/j.ajic.2019.02.028.
3 Kalmeijer MD, van Nieuw-land-Bollen E, Bogaers-Hofman D, de Baere GA. Nasal carriage of Staphylococcus aureus is a major risk factor for surgical-site infections in orthopedic surgery. Infect Control Hosp Epidemi-ol. 2000;21(15)319-323
What is the shelf life of Profend® Nasal Antiseptic Kit?
The shelf life of Profend Nasal Antiseptic Kit is 34 months
Product sizes and specifications are available in the convenient chart below.
| SKU Number | Product Size | Dimensions | Packaging | GTIN | Country of Manufacturer |
|---|---|---|---|---|---|
| X12048 | Swabstick Kit | 2 3/16" x 7/8" x 3 7/8" (Kit) | 4/48 | 20310819008666 | USA |